What is a Lithium Battery and How Does It Work?
Lithium Batteries are a crucial part of modern technology. They power our smartphones, laptops, and electric vehicles. Understanding how they work is essential for grasping their impact on daily life.
These batteries rely on lithium ions to generate energy. The movement of these ions between the anode and cathode creates an electric current. Their lightweight nature and high energy density make them popular. However, lithium batteries are not without concerns. They can overheat or even catch fire.
The growing demand for lithium batteries raises questions. Are we prepared for potential environmental impacts? It’s vital to reflect on the sustainability of lithium mining. As we embrace this technology, let’s consider its future carefully.
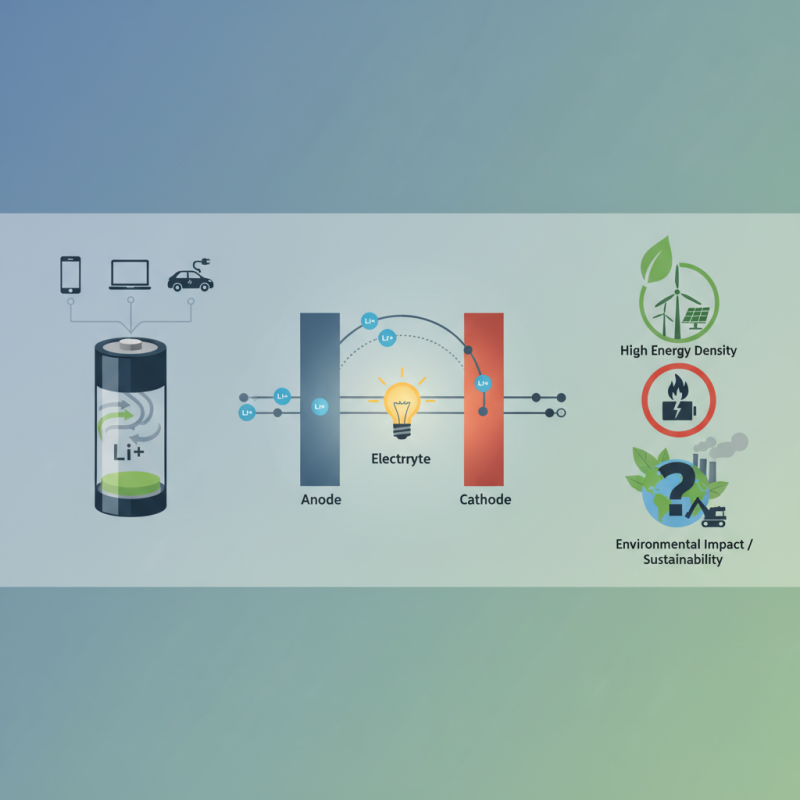
What is a Lithium Battery?
A lithium battery is a type of rechargeable battery that uses lithium ions as the primary component of its electrochemistry. These batteries are lightweight, have a high energy density, and can efficiently store electrical energy. Industry reports indicate that the global lithium battery market is set to grow significantly, reaching approximately $129 billion by 2027. This growth reflects the increasing demand for energy-efficient solutions, particularly in portable electronics and electric vehicles.
Lithium batteries work through the movement of lithium ions between the anode and cathode during charging and discharging. When discharging, lithium ions move from the anode to the cathode, generating electric current. Conversely, during charging, lithium ions move back to the anode. This back-and-forth motion is what powers your devices.
However, there are concerns regarding the sustainability of lithium mining. Reports suggest that lithium extraction can have harmful environmental impacts, including water depletion and land degradation.
Despite their advantages, lithium batteries pose challenges. They can overheat and, in some cases, catch fire if not managed properly. The existing recycling processes are also insufficient. Currently, less than 5% of lithium batteries are recycled effectively. As the number of batteries in use rises, this issue will only become more pressing.
The Chemistry of Lithium Batteries
Lithium batteries are revolutionary energy sources. Their chemistry is rooted in the movement of lithium ions. These ions travel between the anode and cathode during discharge and charging. This process creates a flow of electricity, which powers devices.
At the heart of lithium batteries is a simple yet effective reaction. When a lithium ion leaves the anode, it moves through the electrolyte to the cathode. At the cathode, it combines with other substances, releasing energy. This is where the real action happens. The materials used in these batteries play a critical role in efficiency and lifespan.
Despite their advantages, lithium batteries have drawbacks. They can overheat, leading to safety concerns. Recycling them is another challenge. Many batteries end up in landfills. This aspect needs attention. As the demand for energy storage grows, so does the responsibility to innovate in battery technology. Balancing efficiency and environmental impact is crucial.
The Components of Lithium Batteries
Lithium batteries are widely used due to their efficiency and lightweight nature. Understanding their components helps us grasp how they function. The main parts of a lithium battery include the anode, cathode, electrolyte, and separator. Each plays a crucial role in the battery's operation.
While the technology is impressive, there are concerns. The materials used can be harmful to the environment. Mining lithium can have significant ecological impacts. Additionally, battery life varies based on usage and temperature. Not everyone may understand this complexity. The more we learn about these components, the better we can innovate safe and effective battery technology.
How Lithium Batteries Generate Power
Lithium batteries are a crucial component in our daily lives, powering everything from smartphones to electric vehicles. Understanding how they generate power is essential. At the core, these batteries utilize lithium ions to produce electricity. When the battery discharges, lithium ions move from the anode to the cathode, generating a flow of electricity. This flow powers devices and keeps them operational.
Recent data indicates that the global lithium-ion battery market is expected to grow significantly. Reports suggest a growth rate of over 20% annually. As demand increases, so do concerns about the sustainability of lithium extraction. This prompts a deeper reflection on resources. Are we overly reliant on a finite material? As we explore new technologies, we must balance innovation with sustainability.
Tips: To prolong the battery life, avoid extreme temperatures. Recharge regularly but don’t let the battery deplete entirely. Small habits can ensure better performance. Keep in mind that advancements in technology are also crucial for the environment. Transitioning to alternative materials can be a step in the right direction. Remember that every action counts in creating a more sustainable future.
Applications and Advantages of Lithium Batteries
Lithium batteries are becoming increasingly popular in various applications. Their lightweight design and high energy density make them ideal for portable electronics. According to a recent market report, the global lithium-ion battery market is expected to reach $100 billion by 2025. This surge is driven by demand in electric vehicles (EVs) and renewable energy sectors.
In the automotive sector, lithium batteries are crucial for EV performance. They offer longer ranges and faster charging times compared to traditional lead-acid batteries. A study reveals that using lithium batteries can improve an electric vehicle's range by up to 30%. However, there are challenges. Manufacturing these batteries involves extensive mining of lithium and cobalt, raising environmental concerns.
Moreover, safety remains a concern. Lithium batteries can catch fire or explode if damaged or improperly charged. Reports indicate that nearly 10% of all lithium batteries face safety issues. While their advantages in efficiency and energy storage are significant, the industry must address these risks. Overall, lithium batteries are a double-edged sword, offering great promise alongside serious challenges.
